About Us
Who We Are
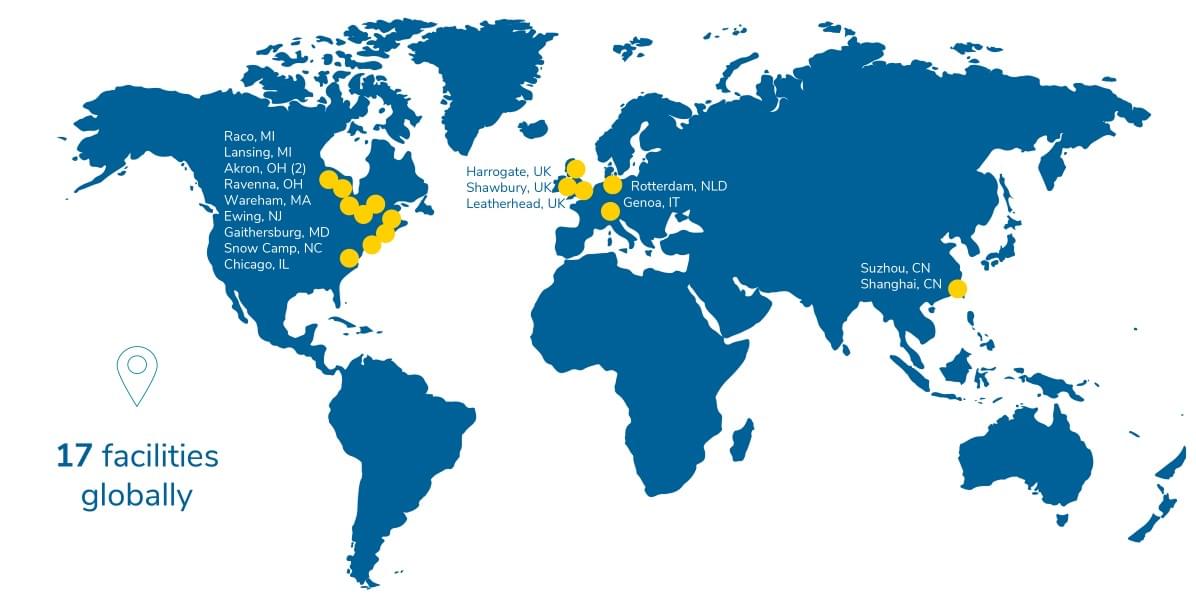
For nearly 100 years, Smithers has been providing accurate data, on time, with high touch to our clients. Founded in 1925 and headquartered in Akron, Ohio, Smithers is a multinational provider of testing, consulting, information, and compliance services. With laboratories and operations in North America, Europe, and Asia, Smithers supports customers in several industries essential to human lives including:
- Transportation
- Life Science
- Food and Packaging
- Materials
- Components
- Consumer Goods
- Dry Commodities
- Energy

Accurate Data, On Time, With High Touch
Accurate Data, On Time, With High Touch
Smithers believes in an ethical, client-focused culture to build trusting relationships. This belief has been integral to Smithers for nearly 100 years.
Client Testimonials
Client Testimonials
See what some of our esteemed clients have to say about their experiences working with Smithers experts and the value that Smithers brings to every project.