In this third of a four-part series, Smithers Study Director, Sidney Bluemink, discusses the varied application of QuEChERSER.
Part 1 discusses the wide-ranging application of QuEChERS and its variants. Part 2 covers the method and application of QuEChERS.
QuEChERSER (Mega-Method)
Traditional QuEChERS has already been around for 20 years. The instrument advancements at the time allowed these methods to be implemented but since then, many QuEChERS methods have remained largely the same while instrument performance has advanced further. Using a QuEChERS method designed 20 years previously with the sensitivity of today’s QqQ, Q-TOF or Orbitrap™ mass spectrometers, greatly reduces the shortcomings of the approach.
Before QuEChERS, typical sample weight to solvent ratios could range from 1:2 to 1:10 and it was common for 50 g of material to be extracted with 100 mL or more of solvent, followed by various labor-intensive shaking, evaporation, partition and SPE steps. With QuEChERS, the typical sample weight to solvent ratio is 1:1 (g/mL) and typically 10 g of commodity was extracted with 10 mL of acetonitrile. This is convenient for a few reasons:
- This setup easily fits into ubiquitous and inexpensive 50 mL polypropylene centrifuge tubes.
- The lower solvent volume allows for more concentrated extracts (and thus lower detection limits) often negating the need to include an evaporation step to concentrate the sample.
- The weight to solvent ratio delivers sufficient extraction efficiency for most applications but note this is problematic for lipophilic analytes in fatty or oily commodities.
The limitation of QuEChERS for lipophilic analytes is solved by the QuEChERSER (
Quick,
Easy,
Cheap,
Effective,
Rugged,
Safe,
Efficient and
Robust) variant, or “Mega-Method.”
The aim of the Mega-Method is to encompass more analytes and more matrices while at the same time reducing even further the use of consumables and chemicals and with fewer method iterations. As with QuEChERS 20+ years ago, advancements in instrument performance allow laboratories to simplify sample workup even more while still retaining method robustness.
One advantage of QuEChERS can also be seen as a disadvantage depending on the context. The single extraction method can include both liquid chromatography (LC) and gas chromatography (GC) amenable analytes. While this is certainly efficient, it can force compromises in analytical performance. An acetonitrile extract can be directly injected onto a GC without too much difficulty but injecting a “strongly organic” sample into a reverse phase LC system can introduce peak shape and ultimately signal to noise issues for mostly polar analytes.
The LC peak shape problem can be overcome by additional steps:
 Mega-Method for LC of hydrophilic analytes
Mega-Method for LC of hydrophilic analytes
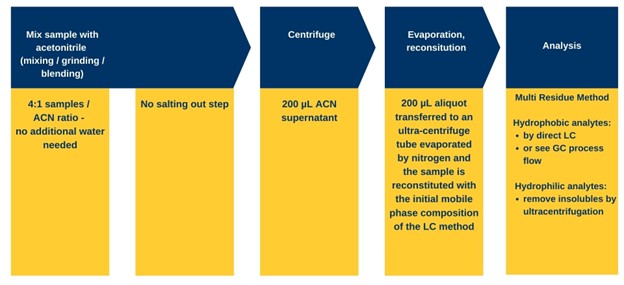
For the analysis step, the reconstituted extract will at this point have hydrophobic matrix and analyte precipitation. For an MRM, the
hydrophobic analytes can be analyzed by GC (see next section), or a separate aliquot of the bulk extract could be directly injected with an LC method. Although the extract would be mostly organic solvent the hydrophobic analytes are not as greatly affected by poor peak shape in reverse phase LC. For
hydrophilic analytes, the precipitate must be eliminated to prevent LC column deterioration. The most effective method is ultracentrifugation to obtain a clean enough extract (filtration can compromise at least a selection of analytes for MRM by either eliminating them or introducing other unwanted contaminants). Using a strongly aqueous reconstitution solvent will eliminate a substantial proportion of the hydrophobic components that would have otherwise fouled the column (1). After centrifugation, the extract is ready for injection.
Mega-Method for GC of hydrophobic analytes
While the LC-focused method can be used for the analysis of hydrophobic analytes, a more suitable design is to reserve the hydrophobic analytes for GC analysis.
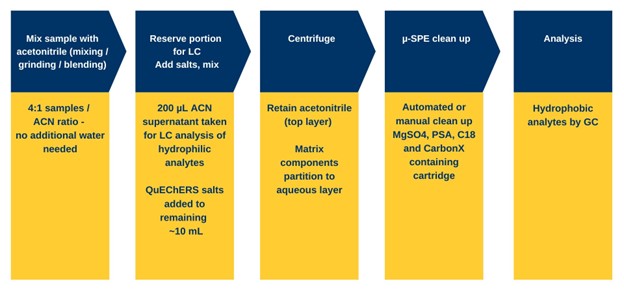
Those familiar with GC analysis will see the problem with injecting a raw 20% aqueous extract sample: contamination of the injection liner, the front of the GC column and the inlet gas lines. Such a raw extract would leave behind non‑volatile matrix components which would significantly increase the number of maintenance cycles and consumable usage of the instrument. Additionally, when water is injected, the liquid expands approximately 1200-fold as it vaporizes, which can overwhelm an injection port liner and cause backflash where the gas lines leading into the inlet are contaminated. This then causes injection carry over and interference with subsequent injections. To reduce the water in the sample, a “salting out” step is included here where the remaining 10 mL of extract is mixed with 2 g of salt mixture. As with QuEChERS, this serves to separate the water and the acetonitrile and eliminates most of the hydrophilic components.
Mega‑Method Considerations
An important consideration of the Mega-Method is the smaller sample weight of 2 g. Care should be taken to ensure that samples are homogenous and representative of the whole sample to be tested. The quality of sample processing is an area that merits greater scrutiny (2,3). Quality equates to achieving smaller particle size (comminution) of the homogenate. The most common method to obtain a homogenous sample is to blend pre-frozen portions of the sample with dry ice using a heavy-duty blender. After the dry ice has sublimated, a frozen, powdery mixture remains which is generally adequate for the kind of sample weights typically used for QuEChERS (10 g). With 2 g sub-samples, it is recommended to blend non-frozen portions of the samples with liquid nitrogen to achieve improved comminution (3).
When developing extraction methods of solids complete extraction of the analyte into the solvent (near 100% recovery) is important. Incomplete extraction can lead to underestimation of the levels of chemicals present in the sample and influence the downstream risk assessment. With the Mega-Method, the improved sample comminution increases the surface area of the particles encountering the extraction solvent. Additionally, the combination of the higher solvent to sample weight ratio and the extraction solvent of choice (acetonitrile: water 4:1 (v/v)) has been shown to be efficient across a variety of analytes and matrices (4).
In the fourth and final part of the series, Sidney will discuss some of the more specialized QuEChERS methods including QuPPe (Quick Polar Pesticides) and QuOil.
Part 1 in this series covers the wide-raging application of QuEChERS and its variants.
Part 2 in this series discusses the method and application of QuEChERS.
Author:.jpg)
Sidney Bluemink
Study Director
Harrogate, UK
References
1. N. Michlig, S.J. Lehotay, A.R. Lightfield, H. Beldoménico, and M.R. Repetti, J. Chromatogr. A 1645, 462097 (2021).
2. S.J. Lehotay and Y. Chen, Anal. Bioanal. Chem. 410, 5331–5351 (2018).
3. S.J. Lehotay, N. Michlig, and A.R. Lightfield, J. Agric. Food Chem. 68, 1468–1479 (2020).
4. S.J. Lehotay and A.R. Lightfield, J. AOAC Int. 103, 584–606 (2020).




.jpg)